
In unveiling the GENErating cost Reductions fOr U.S. Medicaid (GENEROUS) Model on November 6, the Trump administration outlined how it will use federal authority to advance “most-favored-nation”(MFN) pricing as a strategy to reduce drug costs. By channeling the model through the Centers for Medicare & Medicaid Services’ (CMS) Center for Medicare & Medicaid Innovation (the Innovation Center) and structuring it as a voluntary demonstration, the administration also signaled an appreciation for policy lessons learned during President Trump’s first term.
GENEROUS is not the broad, mandatory application of MFN pricing that some anticipated based on the administration’s prior rhetoric. However, as a deliberately designed and legally insulated experiment based on voluntary participation, it could be an appropriate platform for demonstrating the potential of MFN.
The announcement of the GENEROUS Model followed months of escalating White House rhetoric on drug pricing. This included the April executive order (EO) Lowering Drug Prices by Once Again Putting Americans First and a May EO in which President Trump warned that drug manufacturers who failed to offer Americans MFN lowest prices would face “additional aggressive action.”
The administration continued its push towards MFN pricing on July 31, when the White House announced that Trump had sent letters to 17 major pharmaceutical manufacturers — including AbbVie, Amgen, AstraZeneca, Bristol Myers Squibb, Eli Lilly, Johnson & Johnson, Merck, Novo Nordisk, Pfizer, Regeneron, and Sanofi. In the letters, the president called on companies to extend MFN pricing “to every single Medicaid patient,” and warned that failure to do so would prompt the federal government to “deploy every tool in our arsenal” against what he described as abusive pricing practices.
Absent voluntary compliance from manufacturers, the White House could have pursued MFN policy through rulemaking. However, the implementation of GENEROUS through the Innovation Center as a voluntary demonstration specific to Medicaid programs allows for more rapid execution than traditional rulemaking. It also minimizes the potential for substantive and procedural challenges of the sort that prevented implementation of the interim final rule establishing the Most Favored Nation Model in Medicare issued during Trump’s first term.
Speaking at the Northern Virginia Health Policy Forum on November 19, 2025, CMS advisor Dr. Inma Hernandez identified making drugs available to state[1] Medicaid agencies at internationally comparable prices as one of four pillars in the administration’s broader drug pricing strategy.[2] The GENEROUS Model structures this pillar by introducing international reference pricing into Medicaid through supplemental rebates, standardized coverage terms, and state participation agreements.
The model
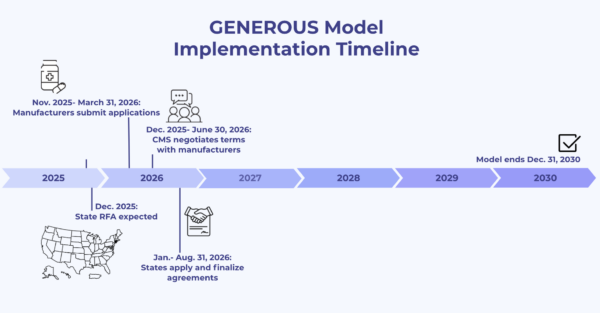
The model outlines terms of participation for states and manufacturers.
Per the Request for Applications from Eligible Manufacturers (RFA), manufacturers wishing to participate must commit to providing MFN-level pricing across their full portfolio of Medicaid-covered single-source and innovator multiple-source drugs.
The model offers states valuable flexibility, including the option to access MFN pricing on a drug-by-drug basis. This ability to target specific drugs may be especially appealing in the context of specific high-cost therapies for which existing rebate programs offer limited savings.
Enrollment will proceed in sequence: manufacturers will apply and negotiate key terms with CMS during the model’s pre-implementation phase. States will follow once the state-level RFA is released and at least one manufacturer agreement is in place. This ensures that states know which products will be included in the program before committing to participation.
Calculating MFN
The cornerstone of GENEROUS is its pricing methodology. The model defines MFN prices using manufacturer-reported average net prices from a basket of peer countries: Canada, France, Germany, Italy, Japan, the United Kingdom, Denmark, and Switzerland. Reported prices must reflect actual net revenues received over the previous 12 months, accounting for all applicable discounts and rebates.
CMS will identify the second-lowest price in this country basket and adjust it based on GDP per capita using a purchasing-power-parity method. The result becomes the MFN benchmark. Using the second-lowest price helps minimize the influence of atypical outlier prices while ensuring the benchmark remains anchored to international market levels.
Translating the MFN Price into Medicaid Rebates
The MFN benchmark is operationalized through a Guaranteed Net Unit Price (GNUP), which establishes the net price target for calculating supplemental rebates. These supplemental rebates are layered on top of existing Medicaid Drug Rebate Program (MDRP) obligations. CMS has indicated in an FAQ that supplemental rebates under the model do not change Medicaid “best price” calculations and therefore do not affect ceiling prices under the 340B program, mitigating risk of unintended pricing consequences across other programs.
A detailed explanation of the pricing methodology is available in the RFA.
Standardizing Coverage Rules
GENEROUS will provide manufacturers streamlined administrative terms. Instead of navigating a patchwork of state-level prior authorization policies and formulary placement decisions, they will negotiate standardized coverage criteria with CMS. Participating states must adopt these terms or implement equally permissive alternatives, ensuring a consistent baseline for coverage across participating states.
This standardization introduces greater uniformity in Medicaid coverage for model drugs while still allowing states flexibility to apply less restrictive policies if they choose. Coverage criteria may be renegotiated if there are updates to FDA labeling or new clinical evidence, enabling states to amend existing participation agreements to reflect current medical guidance. By reducing variability in utilization management and formulary rules, the model has the potential to improve patient access and adherence, particularly for high-cost or complex therapies, while maintaining oversight of Medicaid spending.
The RFA specifies that CMS will work with a contractor to monitor the model’s impact on Medicaid spending, access, and quality of care. This includes tracking pricing outcomes as well as secondary effects such as prescribing patterns, medication adherence, emergency department use, and hospital admissions. The agency will also assess patient-level outcomes, including medication access, utilization management changes, and health system impacts. All model participants will be required to cooperate with these monitoring and evaluation activities.
In this way, GENEROUS functions as a structured experiment rather than a broad policy shift. It offers a means of testing whether international reference pricing can sustainably lower costs while preserving or improving care.
Is Medicaid the Right Test Bed?
The GENEROUS Model assumes that aligning Medicaid drug prices with international benchmarks will yield meaningful savings. However, some observers have questioned whether Medicaid is the ideal vehicle for such a demonstration.
Medicaid already secures some of the lowest drug prices in the country. A 2021 Congressional Budget Office (CBO) analysis found that in 2017, Medicaid’s average net price per standardized prescription was $118, far below the $343 average in Medicare Part D. CBO attributed this difference to the deep rebates built into the MDRP.
MDRP rebates, which include inflation penalties for price hikes, have driven many Medicaid net prices far below list prices. Because CBO did not factor in the supplemental rebates many states negotiate, the program’s actual net prices may be even lower.
This context raises the question as to how much more MFN pricing can save in Medicaid, when individual Medicaid programs may already be outperforming other programs and even peer nations.
States may, however, find the GENEROUS Model particularly useful for newer, high-cost therapies that lack generic competition. For such products, international net prices may fall well below Medicaid’s statutory minimums, making MFN-based pricing a potential source of meaningful savings.
While Medicaid may not be the most obvious testing ground for MFN pricing, it may be the most feasible. With built-in rebate systems and defined coverage frameworks, the program provides a safe space to evaluate the policy’s effects without the risks of legal challenges that might arise from broader implementation.
What we will be watching
Several dynamics will shape whether the model is ultimately expanded. Foremost among these will be manufacturer participation and state engagement. Without a broad level of participation, GENEROUS risks generating inconsistent results with diminished policy relevance.
Another key factor is how manufacturers respond to MFN pricing in the United States. Companies could adjust global pricing strategies to manage exposure to MFN benchmarks, which may influence the net savings achieved within Medicaid.
Observing these interactions will provide insights into the practical challenges of implementing international reference pricing within Medicaid and inform decisions about potential model expansion or adaptation.
Ultimately, the impact of the GENEROUS Model will depend on manufacturer participation, state engagement, and how pricing strategies evolve in response to the model. By monitoring both cost and patient-level outcomes, the model offers a unique opportunity to evaluate whether international reference pricing can produce meaningful savings in Medicaid while preserving access to care. The findings from this experiment will be critical in informing future policy decisions about the broader application of MFN pricing in the U.S.
[1] As used herein, “states” refers to the 50 states, the District of Columbia, and U.S. territories.
[2] According to Hernandez, the remaining pillars are:
- Direct-to-consumer discounts, which allow manufacturers to offer significantly reduced cash prices;
- Prospective MFN launch pricing, intended to ensure that new drugs do not launch in the United States at higher prices than in comparable countries; and
- Revenue repatriation, which would require manufacturers to return a portion of additional overseas revenue linked to U.S. trade policy.